Ozone Oxidation
Automotive Research
 1. OUR CARS PRESENT AN IDEAL ENVIRONMENT FOR BACTERIA. ASTON UNIVERSITY, Anglia 2013/07/20
1. OUR CARS PRESENT AN IDEAL ENVIRONMENT FOR BACTERIA. ASTON UNIVERSITY, Anglia 2013/07/20
2. Surface germicidal effects of ozone for microorganisms AIHA J (Fairfax, Va.)USA 2003 July -August
3. Car indoor air pollution – analysis of potential sources Daniel Müller* Institute of Occupational, Social and Environmental Medicine, Goethe-University, Frankfurt, Germany 2011 December
4. Don’t Drive and Breathe Pollutants Lurk Inside Vehicles BOB WEINHOLD /ehpnet1.niehs.nih.gov/docs/2001/109-9/focus.html 24 Oct. 03
How Does Ozone Work?
 Like chlorine and chlorine dioxide, ozone disinfects by oxidation. This results in a reduction of pathogen populations and disinfected water and produce surfaces. Ozone’s oxidation power is higher than both chlorine dioxide and chlorine.
Like chlorine and chlorine dioxide, ozone disinfects by oxidation. This results in a reduction of pathogen populations and disinfected water and produce surfaces. Ozone’s oxidation power is higher than both chlorine dioxide and chlorine.
The ozone molecule is very unstable and has a short half-life. Therefore, it will decay after some time into its original form O2 Actually ozone is nothing more than oxygen (O2), with an extra oxygen atom, formed by an electric high charge an extra oxygen atom. In nature ozone is produced by some chemical reactions. The special “fresh, clean, spring rain” smell is a result from nature-produced ozone as well as by ozone generators.
Harmful Oxidation by Ozone?
Regarding the ozone oxidation effects, many true facts and findings are listed in various sites and journals, but unfortunately also misleading and false ozone caused oxidation for different materials are published. Material oxidation capability produced by ozone generators does exist but only when using inappropriate ozone (very high) concentration and extended length of duration.
The Mobilsept™ HCG O3 unit`s ozone capacity is specially designated for the disinfection/deodorization treatment with automatically pre-programmed 25 minutes running time. The recommended ¼ yearly use (or even monthly) will not harm any part of the vehicle`s interior.
Oxidation
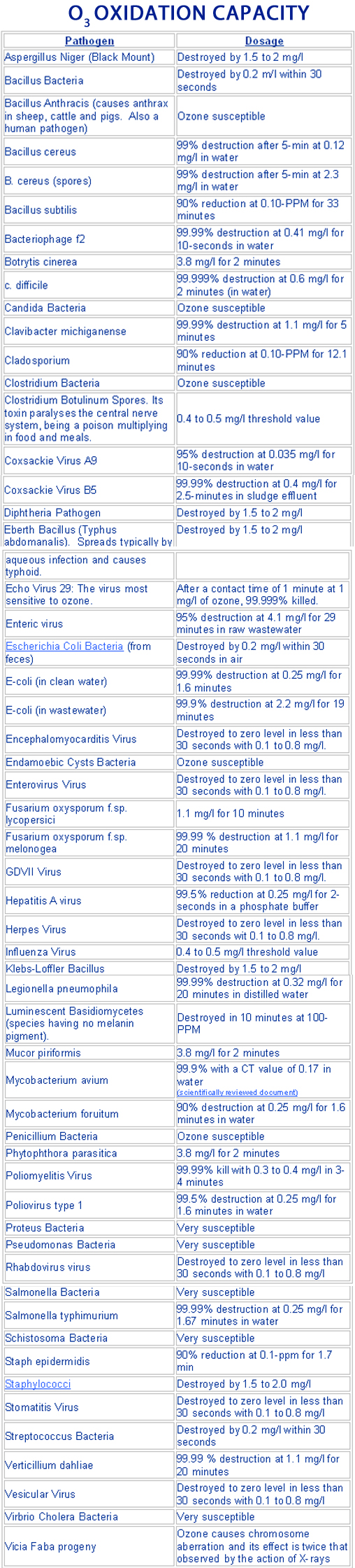
Bacteria are microscopically small, single-cell creatures having a primitive structure. The bacteria body is sealed by a relatively solid-cell membrane. Ozone interact with the metabolism of bacterium-cells, by inhibiting and blocking the normal conditions of the enzymatic control structure. The proper dosage of ozone will destroy the cell membrane, which result to the destruction of the bacteria. Viruses are tiny, independent particles, assembled of crystals and macromolecules, Differently from bacteria, they multiply only within the host cell. In this process changing protein of the host cell into proteins of their own. Ozone kills viruses by dispersing through the protein coat into the nucleic acid core, resulting in damage of the viral RNA. The increased dosage, ozone destroys the capsid, or exterior protein shell by oxidation so DNA (deoxyribonucleic acid), or RNA (ribonucleic acid) structures of the microorganism are affected.!!! The efficacy of ozone below the critical concentration value is little or zero. Above this level all pathogens are will be destroyed. The list below is an all-or-none response and the critical level of the “threshold value”
1-mg/l = 1-PPM